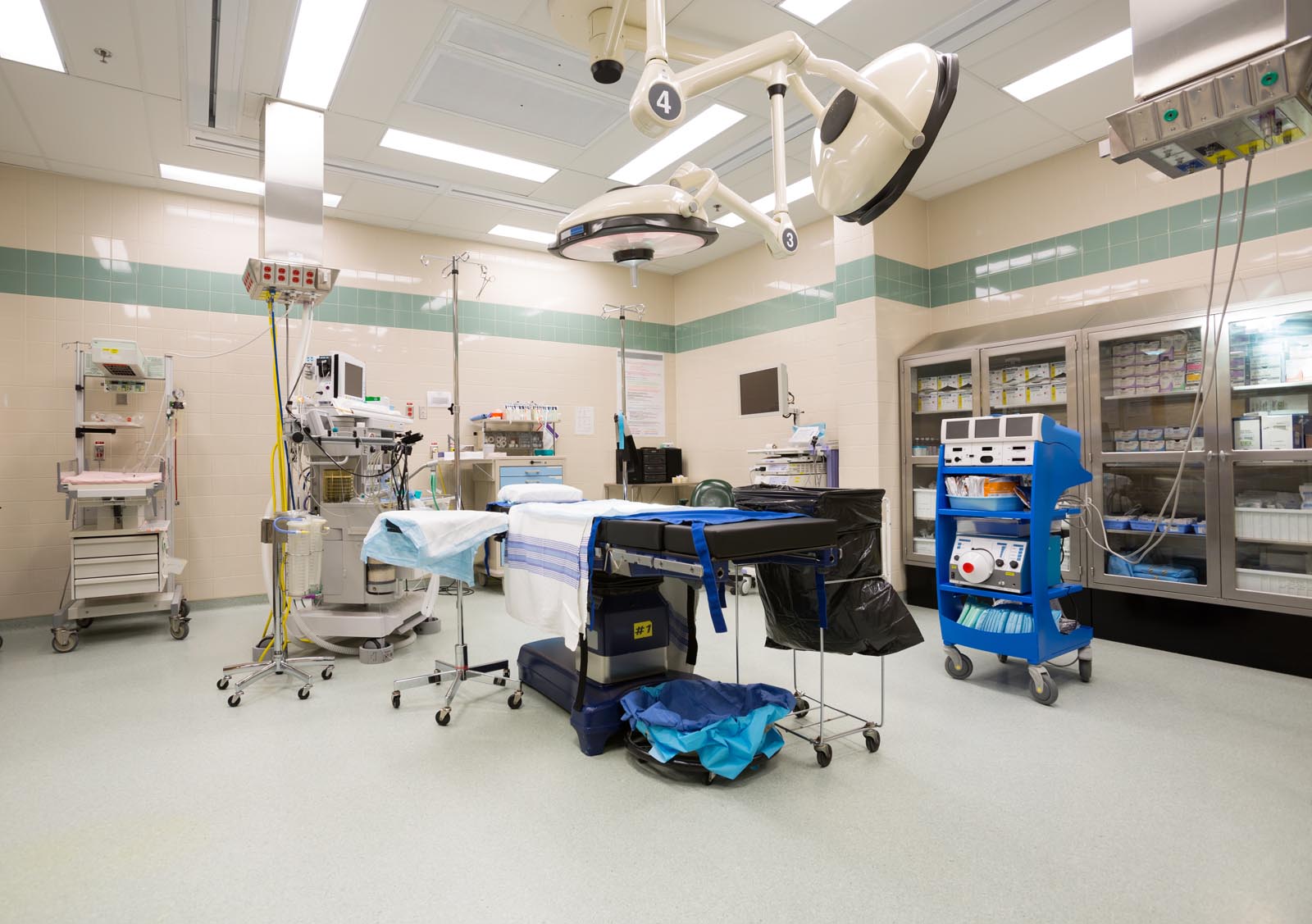
Welcome to Med Val Pharma
Med Val Pharma Limited are a specialist medical and pharmaceutical validation consultancy company.
The company provides validation consultancy to meet international regulatory inspectorate requirements to the Healthcare Bio Medical Pharmaceutical industries. We are specialists in providing a solutions orientated validated compliant approach to achieving budgetary as well as time restricted project completion.
Med Val Pharma Limited have developed an approach based on providing a pragmatic solutions consultancy that is founded on effective customer relationships by understanding both legislative and client requirements. Our success is formulated on understanding our clients’ needs to achieve classification compliance with NHS, Pharma and the Bio-Med industry statutory expectations.
AE(V) Consultancy
As a certified AE (V) by the SVH (Specialist Ventilation in Healthcare) Society, we can work closely with the Trust Senior management to follow fully the code of conduct as prescribed by the AE(V) SVH Society to audit and access ventilation systems within the healthcare environment.
The SVH AE(V) Society member is an accredited body associated with I HEEM, Institute of Healthcare Engineering and Estate Management.
Our consultancy covers a wide range of technical accreditations as follows –
Validation Documentation
Med Val Pharma have established MHRA accredited validation documentation consultancy in setting UP, URS, DQ, IQ, OQ, PQ and VMP validation protocols.
The full documentation package can be put together to meet MHRA / FDA expectations for licensing of the premises.
JACIE accreditation is also a consultancy service provided under the umbrella of Med Val Pharma Limited.
Validation document training and document authorising can be part of the overall package.
With Change Control, Impact Assessment protocols the full validation documentation certification can be provided.